
Consumer Product Development
Spotlight
Company: Polychrome Medical
Industry: Medical Diagnostic Equipment
HQ: Eden Prairie, Minnesota, USA
Webpage:

PolyChrome Medical LLC has been involved in sweat chloride testing for 20 years, manufacturing the CF Indicator™, a qualitative screening test for cystic fi brosis. The CF Quantum® Sweat Test System is PolyChrome’s new quantitative diagnostic test
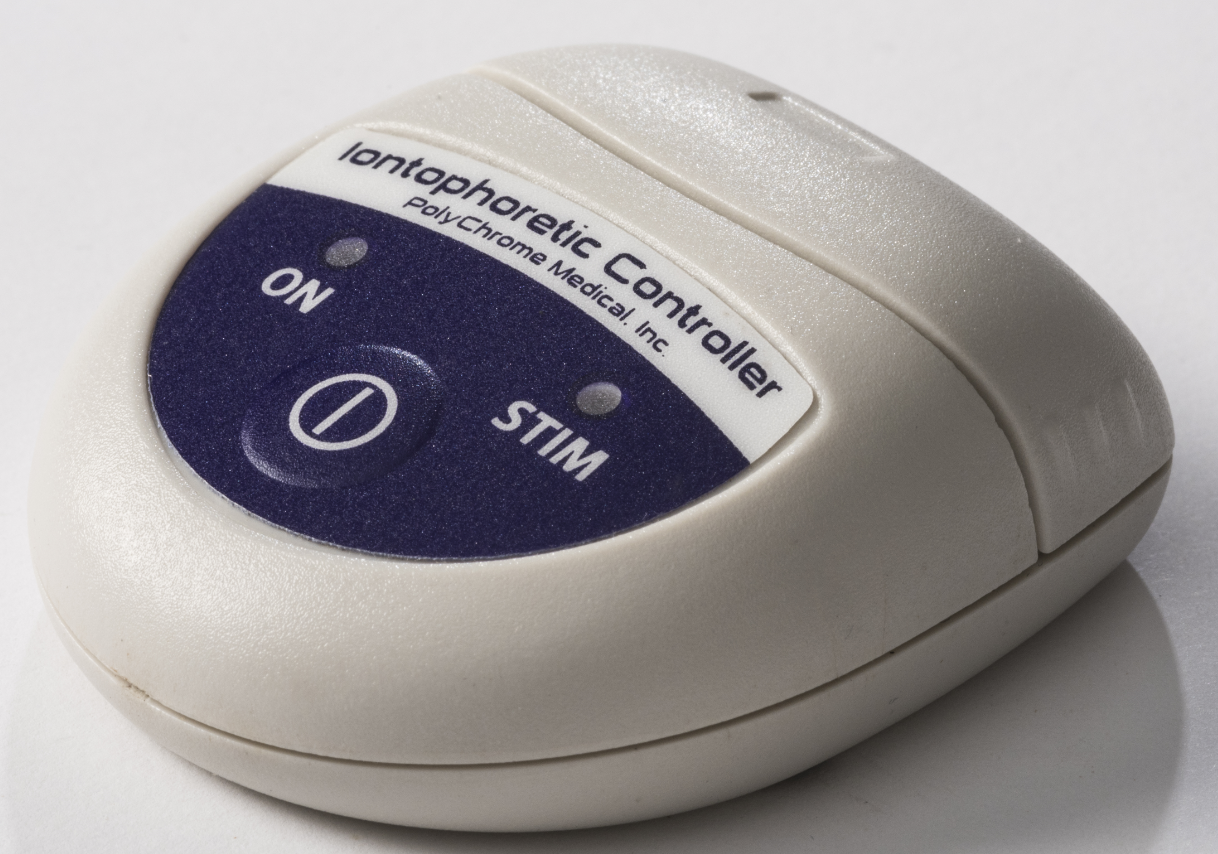
Product/Project Highlights
CF Quantum Controller
Client: Polychrome Medical
- Part of the CF Quantum System to detect cystic fibrosis in infants
- Iontophoresis device – pilocarpine elicits a sweat reaction
- Sweat is gathered onto a patch for testing
- Immediate results, no wait time for diagnosis


Situation:
PolyChrome Medical approached Solid to create a new class II medical device based on a predicate device developed by Medtronic Corporation in the early 1980s. This iontophoresis device drives pilocarpine ions into the patient’s skin in order to elicit a sweat sample for lab analysis. The device delivers an electric current for a precisely limited duration and signals the operator regarding status of the delivery. The Controller needed to be battery-driven, reusable, and snap onto a disposable adhesive pad that contains a set of printed electrodes with hydrogels. The electronics, firmware, enclosure, disposable electrodes and user interface all needed to be designed and developed.
CF Quantum Controller
Client: Polychrome Medical

Process:
Solid participated from the beginning through prototype testing following our Phase Gate Process for product design and development. Involvement included all Industrial Design, mechanical engineering, design and project management. Electrical engineering was conducted by a contracted resource managed by Solid. Solid started by working with PolyChrome Medical to summarise the product requirements, FDA requirements for this type of Class II medical device and then developed multiple conceptual approaches to meet these requirements. Together with our Client, PolyChrome Medical, Solid developed the product architecture, physical form, mechanical functionality and user interfaces for both the re-useable controller and the disposable electrode set. Once the conceptual approach was refined and approved by our Client, Solid completed the detailed design in CAD, conducted detailed design reviews, created engineering drawings, sourced suppliers and solicited proposals for both prototype and production quantities and for production tooling. Solid then coordinated all prototype component moulding, printed circuit build and final programming. Solid was instrumental in determining the strategy and functionality for firmware and electronics.
Solution:
The CF Quantum Controller was born. Along with the disposable electrode set, this device consistantly produces a sweat reaction in infants with minumal discomfort in a short time period. Solid contracted with resources for prototyping and testing of the hardware and software, supported EM testing, supported FDA submission documentation and to date, has supported the initial pilot production build of these devices.


